We provide a comprehensive set of quantitative imaging services for all clinical trials. All services offered are based on the FDA code of federal regulations and guidelines and International Conference on Harmonisation Good Clinical Practice guidelines.
Consultation
We offer trial design and development services as part of our total solution package. Our imaging and oncology experts are always available to you for consultation. Our sub-specialized expert radiologists, who have served in the ACR Appropriateness Criteria-Expert Panel committees, will provide detailed and informative answers to all your questions to meet the needs of your clinical trial goals.
Image Analysis
Image analysis is performed by a highly qualified team of radiologists and imaging professionals who undergo a systematic training and evaluation.
Our imaging services in Oncology include response to therapy assessment for various cancers:
- Breast
- Colon
- Head and Neck
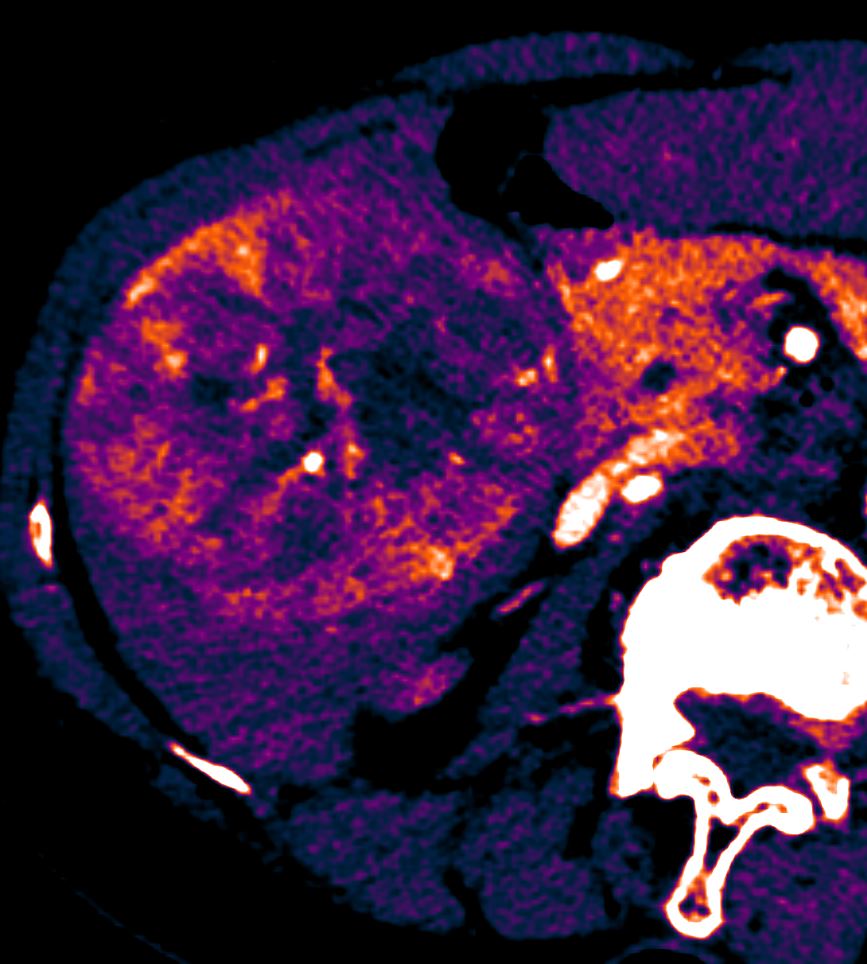
- Liver
- Lung
- Lymphoma
- Pancreatic
- Prostate
- Sarcoma
We provide 2D, 3D, and 4D image analysis capabilities for various study types:
- Computed Tomography (CT)
- Magnetic Resonance Imaging (MRI)
- Positron Emission Tomography (PET)
- Computed Radiography Images
CTI Core Lab makes quantitative measurements of lesions according to accepted techniques and assessment criteria standards such as:
- All versions of RECIST such as:
- RECIST 1.0 and 1.1
- mRECIST
- Immune Related RECIST
- CHESON
- EASL
- Immune Related Response Criteria
- LUGANO
- PCWG3
- PERCIST
- RANO
- Volumetric Size and Density
- WHO
Imaging Protocol Development
Our leading experts in radiology and oncology can exclusively tailor an Imaging Protocol for your clinical trial to satisfy all your study objectives and maximize the effectiveness of image analyses in therapy assessment.
Imaging Biomarker Services
CTI Core Lab is actively supporting efforts to develop and promote novel Imaging Biomarkers for assessing response to therapy in oncology. We can help you to decide which set of Imaging Biomarkers is right for your clinical trial.